Technical Notes on J100 anti-glutathione IgG
1. J100 targets GSH and GSSG. J100 binds to glutaraldehyde (GA) linked to GSH and GSSG via their γ-glutmate amino groups high affinity (high pM range). In conventional mixed aldehyde fixation, GSH and GSSG are linked to the protein matrix (Mx) as GSH-GA-Mx and Mx-GA-GSSG-GA-Mx links. Formaldehyde linking alone leads to mobile methyl-GSH and dimethyl-GSSG species that are lost on sectioning and etching.
In Fig. 1, a 200 nm etched resin section containing rabbit retina fixed in (top) 4% FA buffer versus 4% FA + 0.1% GA (10 mM GA) yields very strong signals in the presence of GA and essentially none in FA-alone. This is due to the loss of methyl-GSH/GSSG.
Fig. 1
2. Can J100 detect glutathionylated protein (GSSP)? This remains an open question. It is possible that J100 binds Mx-GA-GSSP, but this has been difficult to prove.
(i) As shown above, J100 cannot detect methyl-GSSP.
(ii) J100 has never been able to detect GSSPs in Western blots.
(iii) If we assume that J100 can bind Mx-GA-GSSP in tissue, can it be detected? This depends on the intracellular GSSP level. In human red blood cells, the total GSSP content is less than 10% of the available GSH (Giustarini, D et al. 2003 Protein Glutathionylation in Erythrocytes. Clinical Chemistry 49: 327-330). Our measurements of J100 binding in human RBCs report 0.5-0.7 mM total glutathione. A GSSP level of 50 μM would be at the lower limit of our detection with conventional J100 visualization. Further, in Arabidopsis, only 1.1% of glutathione was found to be in the form of GSSP (Dixon DP et al. 2005 Stress-Induced Protein S-Glutathionylation in Arabidopsis. Plant Physiol 138: 2233-2244.) While we can readily detect 1-5 mM glutathione in Arabidopsis cells (see Molecule of the Month: Glutathione) , 1-5 μM GSSP would be a log unit below detectivity of J100 with conventional methods. Even with oxidation stress induced by high levels of tert-butylhydroperoxide, GSSP levels reach only 4.3% of total glutathione, again far below the detection limit for J100. In sum, even if J100 binds GSSP, detecting it is highly improbable.
3. J100 effectively detects total free glutathione. The J100 signal will always be limited by the cellular pool of glutathione and, under normal conditions, by GSH. But even if shifts in the GSH:GSSG ratio occur, the total J100 signal will not change. J100 binding = GSH + GSSG. If J100 cannot detect GSSP, the most we can expect is a small (<10%) change in J100 binding due to GSSP formation. If J100 can detect GSSP, then the J100 signal will always be independent of the oxidation state. However, total glutathione can change via the action of γ-glutamyl transpeptidase, which can lead to a 50% loss in GSH upon activation in some mammalian cells (Allison RD, Meister A 1981 Evidence that Transpeptidation is a Significant Function of γ-Glutamyl Transpeptidase. (1981) J Biological Chemistry 256: 2988-2992.)
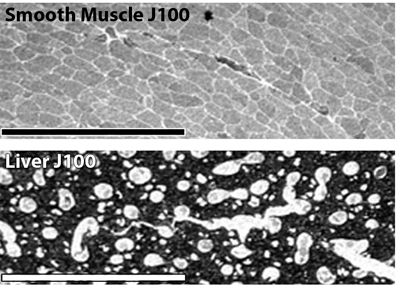
4. J100 quantitatively detects total free glutathione. In immunocytochemical analysis of thin sections, J100 detects over intracellular levels of 0.1-10 mM. Silver-visualized IgGs have a first-order dynamic range and concentration (C) dependence, with binding or displacement on the 2D section surface simply and accurately modeled from digital pixel values (PV) with a saturation function:
PV/PVMAX = C/(C+K)
where PMAX is the maximally measured signal, PV the measured signal in a cell, C the unknown concentration and K the half-saturation function. The "Hill" coefficient is 1. Below (Fig. 2) is the J100 anti-glutathione binding curve calculated from pure GSH and GSSG displacement. J100 does not discriminate between GSH and GSSG. Using this function, we pin calibrated signals to artificial GSH standards. These methods compare well to published biochemical data. Silver images of liver and smooth muscle J100 signals (Fig. 3 below yield levels of 6 and 0.660 mM respectively, which matches calculated values from biochemical measurements well. Published GSH values for rat liver convert to 6.15 mM.(Mercier S, et al. 2002 Chronic inflammation alters protein metabolism in several organs of adult rats. J Nutr. 132:1921-8.) The published GSH values for porcine gastric smooth muscle convert to 0.625 mM (Colpaert EE et al 2002 Influence of antioxidant depletion on nitrergic relaxation in the pig gastric fundus.Br J Pharmacol. 135:917-926.)
Fig. 2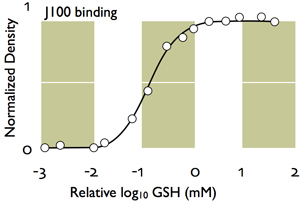
Fig. 3